Supercritical Water Oxidation in Waste Water Treatment
Introduction
SCWO is a one-step wet oxidation method of converting organic materials into carbon dioxide, water, and inert mineral solid residue which is on based on the waste being treated. The method is highly successful and can handle a wide range of wet materials without the need for dewatering. It is an innovative oxidation reaction with immense potential for treating a variety of organic wastes, particularly condensed wet wastes in slurries resembling sludges, bio-solids, agricultural wastes, chemical wastes with unmanageable chemicals like polyfluoroalkyl substances (PFAS), and perfluoroalkyl and several others. SCWO depends on the distinctive reactivity and dispersion qualities that arise whenever an aqueous waste stream is raised over the critical point of water i.e. 218 atm pressure and 374 degrees Cecius. Supercritical water has gas-like transport capabilities and non-polar solvent-like properties. Because oxygen is completely soluble in supercritical water, the organics are oxidized to carbon dioxide, clean water which can be recycled, and even certain non-leachable inorganic salts.
To make SCWO cost effective, the energy from oxidation process is recaptured and then used to heat the influent fluid in portion, with the extra heat turned to power. SCWO reactors can function auto-thermally, requiring no external heat flow, based on the composition of debris in the feedstock. SCWO systems are quite modest when compared to other techniques because typical response time is in the range of 2-10 seconds. There are no toxic by-products like Sulphur oxides (SOx), nitrogen oxides (NOx), carbon monoxide (CO), or odours produced throughout the process. Organic nitrogen and ammonia in the waste being treated are typically transformed to nitrogen gas. Whereas phosphorous precipitates like phosphates could be recaptured. When halogen-containing pollutants are processed, halogen-carbon bonds are often disrupted, and halide anions are released into solution. The SCWO technique for wastewater recovery is based on supercritical water’s unique physical features. Water’s density, ionic product, and dielectric constant all decrease as it approaches its critical point, therefore supercritical water behaves as a non-polar solvent with great diffusivity and great transport characteristics. As a consequence, non-polar organic molecules and gases, such as oxygen, become totally soluble with supercritical fluid. In room temperature, dielectric constants of common organic solvents are being shown below in the picture.

The polarity of water changes with temperature and pressure. Supercritical water (SCW) also has perfect solubility with oxygen, resulting in a homogenous basic mixture, making it an excellent medium for organic oxidation. The response of SCWO can sometimes be heterogeneous whenever the biological matter is a hard material or even in the case of distributed catalytic SCWO.
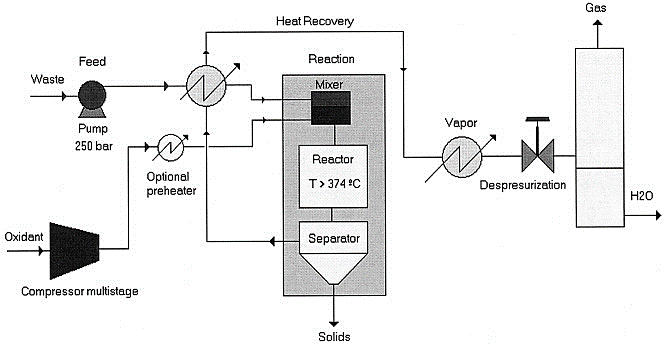
Treatment Process
SCWO comprises of four main phases elimination of organic wastes
- Feed preparation and pressurization
- Reaction
- Salt separation
- Heat recovery and depressurization
1. Feed preparation and pressurization
The oxidant plus effluent streams make up the SCWO reactor’s feed. The performance of the system is regardless of the oxidant because oxygen from the air, hydrogen peroxide, or liquid oxygen can all be employed. Economic considerations govern whether air or oxygen is used as an oxidant. Compressed oxygen is far less expensive than compressed air. They nevertheless add significant material cost. While using hydrogen peroxide at bench-scale operations may be useful, its commercial applicability is constrained due to its high cost. Organic substances in an aqueous media are injected into the reactor at operating pressures (P > 22.1 MPa). If the heat capacity of the debris is insufficient, the thermal efficiency of the feed is regulated to an adequate upper oxidation operating temperature of about 700–750°C by diluting adding water or adding fuel. The feed could be warmed through heat exchange with hot discharge removed from the reactor if the heating value of aqueous effluent is too minimal.
2. Reaction
Whenever the oxidant and organic waste sources are combined with heated agents, an exothermic reaction occurs. The heat generated by oxidation reaction heats the reactant to the operating temperature, where all organic matter is quickly oxidized. The reactor is built to withstand the oxidative conditions and rigorous operating conditions. The reaction temperature, residence duration, oxidant content, and pressure are the most important operating factors.
Reaction Temperature: When the temperature of reaction rises, the efficiency improves and the amount of time required for total reagent oxidation decreases. At temperatures about 650°C, the required residence time for full conversion is 50 seconds, regardless of the pollutants being treated.
Oxidant Concentration: Cumulative removals are calculated using the stoichiometric quantity of oxidant.
Residence Time: Subject to the boundary temperature, residence times can range between a few seconds to one minute.
Operation Pressure: Once the pressure is over the critical pressure of the water (22.1 MPa), increasing the pressure does not enhance conversions. The conversions diminish at lower pressures89, and if the reaction temperature goes up enough, the negative effect of pressure could be mitigated.
3. Salt separation
Because of the low salt absorption, SCWO operations finally produce permanent solid particles. Device fouling, clogging, and erosion can all be caused by these particulates. Despite this, the poor solubility of salt in supercritical conditions allows them to be extracted by a solid–fluid distinction, such as with hydro-cyclones or filtration, allowing valuable products to be recovered. Microfiltration methods are used when a high level of purification in the filtrate is required, but they have drawbacks like corrosion, mechanical placement, erosion, process control and fouling. All of these approaches for retrieving solids at the reactor’s output are only effective whenever the solids need not prefer to adhere to the reactor’s wall. It can occur if the solid isn’t sticky or if the reactor has a system for cleaning solids from the walls.
4. Depressurization and Heat Recovery
The reaction’s gaseous products and supercritical water exit the reactors at a pressure of more than 23 MPa and a temperature of roughly 650°C. This stream should be chilled and depressurized to room temperature before being split into two phases: a gas phase and a liquid aqueous phase. It is feasible to use latent heat of the goods to warm dilute aqueous discharges up to operating condition using a heat exchanger for dilute aqueous wastes with moderate heat of reaction. The amount of organic matter required is determined by the amount of heat delivered. Feeds with a thermal value of 930kJ/kg can provide a discharge with enough energy to heat the feeds at room temperature to 400°C plus produce enough electricity to power the high-pressure pumps and air compressor. Excess thermal energy in the waste could be used to produce steam for outward usage, generate power, or meet the high-temperature thermal needs of manufacturing applications.
Power generation in larger-scale systems could assume the shape of energy production via direct expansion of reactor products via a supercritical steam generator. Such a mechanism would’ve been capable of producing far more energy than is needed for oxygen pumping, air compression, or feed pumping. Energy efficiency of 37 percent with vapor at 30 MPa and 650°C were attained in a theoretical simulation of energy production from coal oxidation by SCWO.
Commercial Applications
Several American companies are now seeking to commercialize supercritical reactors for the destruction of toxic waste. A reactor design that can withstand corrosion and fouling below supercritical conditions is required for widespread commercial implementation of SCWO technology.
There are a number of services SCWO uses in Japan, including one by Organo for the treatment of halogenated effluent. Hanwha has constructed two commercial-sized units in Korea.
The SCWO technology is used for treating waste chemical catalyst to reclaim the precious metal. The AquaCat was commercialized in Europe by Chematur Engineering AB of Sweden. The unit has already been constructed for Johnson Matthey with in UK. It is Europe’s sole commercial SCWO unit and the world’s biggest SCWO machine; with a capacity of 3000 l/h. SCFI Group (Cork, Ireland) has purchased Chematur’s Super Critical Fluids innovation and is aggressively commercializing the Aqua Critox SCWO technology for the treatment of sludge, such as de-inking effluent and wastewater sludge. Many long-term trials on these applications have been conducted, and the distillate after the SCWO procedure is very well adapted for repurposing – in the case of de-inking waste as paper filler or sewage sludge as phosphorous and coagulant – especially with the high destruction effectiveness of 99.9%. In Cork, Ireland, the SCFI Group operates a 250 l/h Aqua Critox experimental facility.
Turbo Systems Engineering in California, USA is focusing on renewable energy applications with their proprietary transpiring wall SCWO reactor.
Aquarden Technologies in Skaevinge, Denmark manufactures modular SCWO systems for the elimination of toxic wastes in industrial waste streams like PFAS, pesticides, and other troublesome hydrocarbons. Aquarden also uses SCWO to remediate hazardous energy pollutants and chemical contamination where a comprehensive SCWO system has been in operation in France for Defence Industry for several years.
Scwo’s Limitations
There are three main reasons why SCWO has not yet become a widely used waste treatment technique:
- Unembellished reactor corrosion induced by acids generated during processing of organics involving halogens, sulphur, or phosphorous, for example.
- Serious reactors get clogged due to precipitated salts at extreme temperatures and low densities.
- Cost analyses, particularly for the build-up of SCWO plants to an industrial level, are difficult to do due to a paucity of experimental observations.
Possible Solutions
Salt-free Effluent Streams
In tube reactors, salt-free effluent streams can be rapidly oxidized. Organics having solely C, H, O, and N, on the other hand, do not produce severe corrosion even when used for longer periods of time. For such wastes, no particular reactor framework is needed. The reactor must be adjusted due to the possibility of corrosion difficulties in the cool-down phase induced by acids generated during oxidizing organics containing additional hetero-atoms. The supercritical sample solution is quenched with a cooler NaOH solution in a revised SCWO method which results in an alkaline, sub-critical mixture. It should be noted that defining a pH value for extreme temperatures is difficult.

Another approach for reducing rusting exists. The existence of a discharge with both a high temperature and a high density at same moment is prevented in this scenario. In contrast to traditional SCWO plants, in which the supercritical solution gets cooled first, then expanded (method A), the sub – critical solution is expanded adiabatically first (way B). After an adiabatic expansion from 600°C to 510°C and 25 MPa to 1 MPa, the temperature drops to 510°C. The resulting low-density hot gas is much less corrosive, and flue gas scrubbers are fully aware of its corrosively. Because of the low mechanical force in this region of the reactor, a less costly design and the use of less pressure-resistant components is possible. The gas is then cooled to its normal temperature.
Waste Discharges Comprising Salt
Waste streams containing excessive salt concentrations will inevitably lead to the clogging of all types of reactors. Other methods should be used to treat such wastes. The unique reactors listed above may be able to completely treat wastes only with low concentration levels of salts, but long-term experiments are required to confirm their applicability. These reactors will undoubtedly raise expenses. It should be determined whether salt separation in a first phase accompanied by SCWO in a tube reactor is a more cost-effective treatment method.
Waste Water Treatment Frequently Asked Questions
1) What is oxidation in wastewater treatment?
Chemical oxidation is used in water and wastewater engineering to transform putrescible polluting chemicals into safe or stabilized products. Natural waters undergo chemical oxidation processes, which are a key mechanism in the surface waters’ natural self-purification.
2) What is super critical water?
Supercritical water is created when ordinary water reaches 373 °C and 220 bar. The “fourth state” of a material might be conceived of as being “supercritical.” It appears to be like a vapour but is actually neither a solid nor a liquid nor a gas. Therefore, visualize supercritical water like visualizing boiling water on a stove.
3) What is oxidation tank?
Large, shallow ponds called oxidation ponds-also known as lagoons or stabilisation ponds—are used to purify wastewater by the action of sunshine, bacteria, and algae.
4) What are the types of oxidation?
Oxidation can be categorized in to three types:
- Spontaneous oxidation.
- Rapid Oxidation.
- Slow Oxidation.
5) What happens when water is oxidized?
Water is converted to oxygen gas and hydrogen ions at the anode. Water is converted to hydrogen gas and hydroxide ions at the cathode.